Last Updated on January 28, 2026 by Don Gerig, RDCS
Back in July 2013, one of the revisions to the IAC Standards and Guidelines included “the structural and electrical integrity of the transducer must be checked between each use, using an ultrasound transducer leakage tester.” This revision became effective December 31st, 2015. That’s great, but why do we have to do this? What is a leakage tester? How does it work? Where can I get one?
In this article, we’ll review why TEE probe leak testing is required, when it must be performed, and how to integrate it correctly into your reprocessing workflow to support patient safety and accreditation compliance.
What Is TEE Probe Leak Testing?
TEE probe leak testing is a process used to evaluate the electrical and structural integrity of a transesophageal probe before it undergoes high-level disinfection and before it is returned to clinical use.
The test is designed to detect:
- Breaks in the probe’s outer insulation
- Internal fluid intrusion
- Electrical leakage that could result in patient or operator injury
These defects are often invisible during routine visual inspection, which is why leak testing is required in addition to visual checks.
Why Leak Testing Matters
1. Patient Safety
TEE probes are used in close proximity to the heart and major vascular structures. A compromised probe can allow unintended electrical discharge, creating a potential risk for patient injury during the exam.
2. Infection Prevention
Small breaches in the probe sheath allow fluids and microorganisms to enter areas that cannot be adequately disinfected. This undermines the effectiveness of high-level disinfection and increases the risk of cross-contamination between patients.
3. Equipment Protection and Cost Control
Early detection of probe damage helps prevent catastrophic probe failure. Given the high replacement cost of TEE probes, consistent leak testing can significantly reduce unnecessary equipment loss.
Accreditation and Regulatory Expectations
IAC Standards and Guidelines expect facilities to demonstrate that TEE probes are inspected and leak tested between every use and that results are documented.
Under current IAC standards, leak testing is considered part of the routine probe reprocessing and quality assurance process.
HOW DO I PERFORM A LEAKAGE TEST?
Thankfully, this procedure is simple and can easily be made an automatic process in your lab.
Overview
Step One:
The first step is to purchase the vendor specific leakage tester for your TEE probe.
Step Two:
Once you have the equipment you can easily implement the use of it into any step of your current cleaning protocols. Remember, testing just requires that the probe to be sitting in a liquid to complete the electrical leakage test. You can test the probe while it’s soaking in the enzymatic cleaner OR while it’s soaking in water.
Step Three:
Plug the connector port into the Leakage Tester and place the electrode from the Leakage tester into the water.
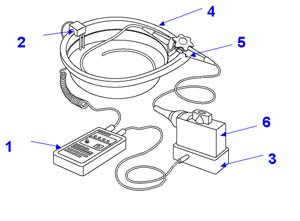
(1) Basin (2) TEE probe with only the shaft immersed and the handle safely outside the basin (3) Transducer Port Plugin (4) Leakage Tester (5) Dual Electrode
Formal Procedure
So now you understand the 3 steps involved to implementing Leakage Testing the next step would include writing a formal policy and procedure and training staff. Here is an example of how your policy can address checking the structural and electrical integrity of the transducer.
Electrical and Structural Testing Procedure
- Pre-clean the TEE probe according to your manufacturer’s guidelines.
- Inspect the TEE probe for signs of damage:
- Lens: Separating from housing, multiple holes, or swollen
- Connector: Bent pins, dents in housing, dirty
- Bending rubber (Flexible shaft): Contaminated or stained, holes and most common, bite marks
- Cable: Pulled out, strain relief, tears, scratches, faded labeling
- Fill the TEE basin (or cylinder, whatever container you soak your probe in) to the level indicated with a saline or enzyme solution that will conduct electricity.
- Place just the Flexible shaft of the TEE probe into the enzymatic solution/saline bath, fully immersing the shaft BUT NOT THE control housing (handle), cable or connector housing
- Place the tester’s dual electrode at least one inch into the enzymatic solution/saline bath.
- Perform conductivity test: Turn test button on. Move switch to Conductivity and click test button again.
- Green light means passed.
- Red light means failed. If conductivity fails, make sure that electrodes are at a depth of 1 inch and everything is firmly connected. Try again. If conduction test fails, do not attempt leakage test. Notify Biomed and or troubleshoot per manufacturer instructions.
- Only if Conduction test passes: Perform leakage test. Move switch to leakage and click test button.
- Green light means passed.
- Red light means failed.
- If Leakage test fails: check all equipment and try again to ensure it wasn’t a procedural error. If Leakage test continues to fail remove the probe from patient care and immediately notify Biomed and or the field engineer.
TEE Leak Testing Case Study
We interviewed the biomedical manager at a hospital in South Florida. He implemented electrical testing three years ago in their facility.
When and why was electrical testing implemented?
The facility was constantly fixing or replacing TEE probes and he realized that damage to the probe was occurring before symptoms of the damage could be seen. This included tiny bite holes undetectable by the eye that after multiple soakings this left the TEE probe unusable. After replacing their probes three times, their warranty was no longer valid and they had to pay $14,000 to replace the probe. A month and a half later it happened again so they implemented electrical testing.
How was testing implemented?
The biomed manager’s goal in addition to ensuring compliance was to make the testing a mindless task, that would be completed automatically. The leak tester they had was complex and not very easy to use and the hospital uses TEE transducers from multiple vendors. He mounted the tester in the sterilization room and modified the sterilization tube to sit on the electrical connector. He also purchased a universal adaptor for the tester that allows many different vendor probes to be tested on it. Now, the tester just has to connect the transducer to the universal converter, dip the tip of the probe into the modified sterilization tube full of water and run the test. The tester is immediately notified if it is “Pass” or “Fail.”
Key Takeaways
- Manufacturer instructions should always guide testing technique
- TEE probe leak testing is a required safety step between every patient use
- Visual inspection alone is not sufficient to detect probe damage
- Leak testing protects patients, staff, and expensive equipment
- Clear documentation supports accreditation readiness
Strengthen Your Lab’s Safety and Compliance With Expert Support
Ensuring your TEE probe leak testing process is reliable and documented is just one piece of accreditation readiness. From full accreditation consulting to quality improvement systems and training, CardioServ provides the expert support echocardiography labs trust to streamline compliance and improve imaging quality.