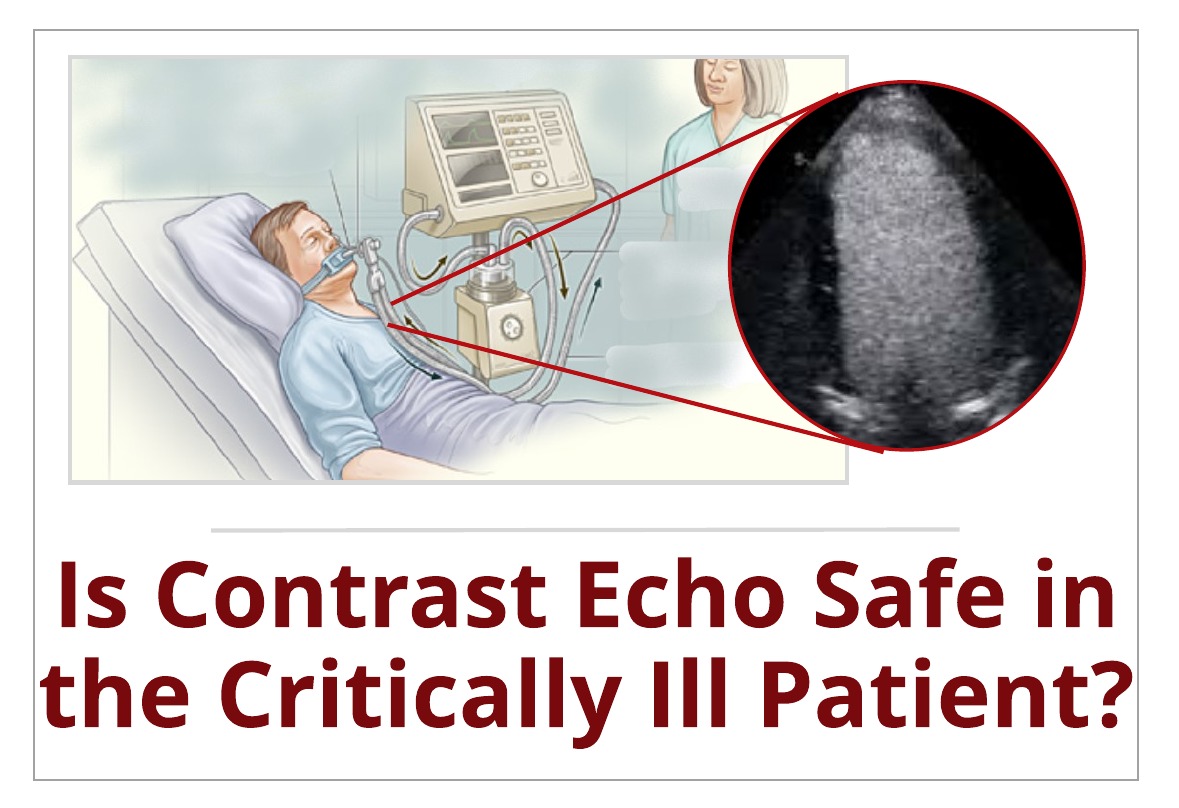
“Isn’t there a black box warning for contrast?” “I don’t feel comfortable giving contrast to my ICU patient” “The patient has acute coronary syndrome, I don’ think you should give contrast” These are just a few of the responses you may face from an ICU nurse or critical care staff in response to your decision to perform a contrast echocardiogram. So what’s the deal? Is contrast echo safe for the critically ill patient? This week we will review the latest data and recommendations regarding the use of contrast in both the critical care setting and the emergency department setting by reviewing the recently released updated guideline paper from the ASE: Clinical Applications of Ultrasonic Enhancing Agents in Echoardiography: 2018 American Society of Echocardiography Guideline Update.
In case you missed it, check out our last article on contrast echo that discussed the new LV volume cut-off values for contrast echo.
HISTORY OF THE BLACK BOX WARNING FOR CONTRAST ECHO
So why the concern about whether contrast echo (Ultrasonic Enhancing Agents) is safe for the critically ill patient? If you remember, back in 2007 the FDA in the US placed a black-box warning on contrast products that included contraindications for certain disease-states. Most of the diseases listed as contraindications were the very reasons that patients were in the ICU! These included things like acute myocardial infarction, acute coronary syndrome, respiratory failure, etc. In addition to the long list of contraindications a mandatory 30 minute monitoring period was placed on all patients.
Following this, in 2008, a number of investigator-initiated studies began evaluating the safety of contrast. The initial results and a plan to complete additional studies resulted in the FDA removing the contraindications and reducing the mandated monitoring to a subset of patients. In 2011, when all the final results were in, the FDA once again updated the DEFINITY product label and acknowledged that serious adverse reactions were “uncommon” and “occur within 30 minutes of administration”. The findings from the registry of 1053 patients which reported no deaths or serious adverse reactions, was added to the label along with the statistic that “these reactions are unlike to occur at a rate of more that 0.3%”. The findings from the pulmonary hemodynamic study was also added that reported no clinically significant changes in pulmonary or systemic hemodynamics. The previous monitoring period for patients with pulmonary hypertension or “unstable cardiopulmonary conditions” was removed.
IS CONTRAST ECHO SAFE FOR THE CRITICALLY ILL PATIENT?
So we have walked down memory lane and reviewed the history of the black box warning for contrast and although the disease-state contraindications were removed from the label all current echo contrast products still contain warnings for the risk for serious cardiopulmonary reactions for a certain subset of patients (acute myocardial infarction, acute coronary artery syndromes, worsening or unstable congestive heart failure, or serious ventricular arrhythmias). So where does this leave us in determining the safety of contrast echo in the ICU? The patients in ICU are often on ventilators, unable to get into the best position to optimize imaging and may even have bandages covering parasternal or apical windows. These are the very patients that have technically limited studies and need contrast in order to better visualize endocardium!! Luckily we have an answer for this based on the research over the past decade.
CONTRAST SAFETY STUDIES: REDUCTION IN MORTALITY FOR CONTRAST PATIENTS!
The safety of our patients is the upmost concern and however great a product may be, at the end of the day we need data and statistics to support the use or non-use of anything we give our patients. So lets review some of the results from the contrast safety studies.
One study matched 16,217 critically ill patients who had received contrast with 16,217 critically ill patients that had not received contrast. “At 48 hours, mortality was significantly lower in the contrast echocardiography arm (1.7% vs 2.5%)” (ASE, 2018)
“A large meta-analysis of these investigator-initiated studies, including 5,078,666 control patients (undergoing echocardiography without a contrast agent) and 211,162 patients who received [contrast] an UCA, demonstrated a significant reduction in mortality for patients undergoing contrast-enhanced echocardiography” (Patil & Main, 2012). During this analysis all databases from inception through 2009 were reviewed for cases of mortality and reported MI.
- “The incidence of death in the contrast group was 0.34% (726 of 211,162 patients) compared to 0.9% (45,970 of 5,078,666 patients) in the noncontrast group
- The reported incidence of myocardial infarction in the contrast group was 0.15% (86 of 57,264 patients) compared to 0.2% (92 of 44,503 patients) in the noncontrast group.
- In conclusion, the cumulative evidence has suggested that the use of contrast agents for echocardiography is safe and not associated with a greater incidence of myocardial infarction or and mortality” (Khawaja et al. 2010).
SUMMARY
Currently contrast echo still carries a warning for the risk for serious cardiopulmonary reactions for a certain subset of patients (acute myocardial infarction, acute coronary artery syndromes, worsening or unstable congestive heart failure, or serious ventricular arrhythmias). Many large studies have demonstrated that contrast echo is safe and not associated with greater incidence of MI and/or mortality. Studies have even reported a reduction of mortality for patients that receive contrast echo. Although there is no direct causitive role in the reduction of mortality with the administration of contrast it is theorized that the use of contrast allows for a more accurate diagnosis of critically ill patients which results in the patient receiving lifesaving treatment in a timely manner.

Judith Buckland, MBA, RDCS, FASE
Stay Connected: Facebook, Twitter, Instagram, LinkedIn
References:
American Society of Echocardiography: http://asecho.org/wordpress/wp-content/uploads/2018/03/UEA-Guideline.pdf
DEFINITY: http://www.definityimaging.com/pdf/Definity%20US%20PI%20515987-1017.pdf
Khawaja OA, Shaikh KA, Al-Mallah MH, Meta-analysis of adverse cardiovascular events associated with echocardiographic contrast agents, Am J Cardiol, 2010;106:742–7. Crossref | PubMed
Main ML, Hibberd MG, Ryan A, Lowe TJ, Miller P, Bhat G. Acute mortality in critically ill patients undergoing echocardiography with or without an ultrasound contrast agent. JACC Cardiovasc Img 2014;7:
408.
Patil, H., & Main, M. (2012, January 03). The History of Product Label Changes for DEFINITY® in the US. Retrieved from https://www.uscjournal.com/articles/history-product-label-changes