Last Updated on April 6, 2026 by Don Gerig, RDCS
Hypertrophic cardiomyopathy (HCM) is more common than most sonographers realize, and it’s missed more often than anyone would like to admit.
In fact, according to ASE, HCM may affect as many as 1 in 200 patients, yet a large percentage remain undiagnosed for years. Many of these patients are already being scanned in everyday echo labs. The challenge isn’t access, it’s recognizing what we’re already seeing. And that’s where we as clinicians come in.
In a recent CardioServ webinar featuring Dr. Ronald Wharton and Margaret Kurnides, ACS, RDCS, FASE, we explored real-world case examples and best practices for evaluating HCM in the echo lab.
The goal of this article is simple:
To help you recognize HCM earlier, scan more intentionally, and avoid the common pitfalls that lead to missed or delayed diagnosis.
This article is part 1 of a 3-part series focused on improving how we evaluate hypertrophic cardiomyopathy in the echo lab.
- Part 1: Recognizing HCM and understanding its key echocardiographic features (this article)
- Part 2: Differentiating LVOT obstruction from mitral regurgitation
- Part 3: Using goal-directed Valsalva to uncover dynamic obstruction
Why HCM Is Often Missed
One of the most important realities about HCM and echo is this:
Many sonographers believe they’ve never come across HCM, when in reality, they almost certainly have, but just didn’t recognize it.
HCM isn’t limited to specialized imaging centers. It presents in all types of echo labs, often appearing with more familiar findings, such as:
- LVH
- Aortic stenosis
- Mitral valve prolapse
- Hypertension
Because these findings often overlap with other common conditions, HCM can easily be overlooked or misinterpreted.
Symptoms are frequently nonspecific, which can delay further evaluation. As a result, patients may go as long as 3–5 years before receiving an accurate diagnosis.
Key Echocardiographic Findings of HCM
Start With Pattern Recognition
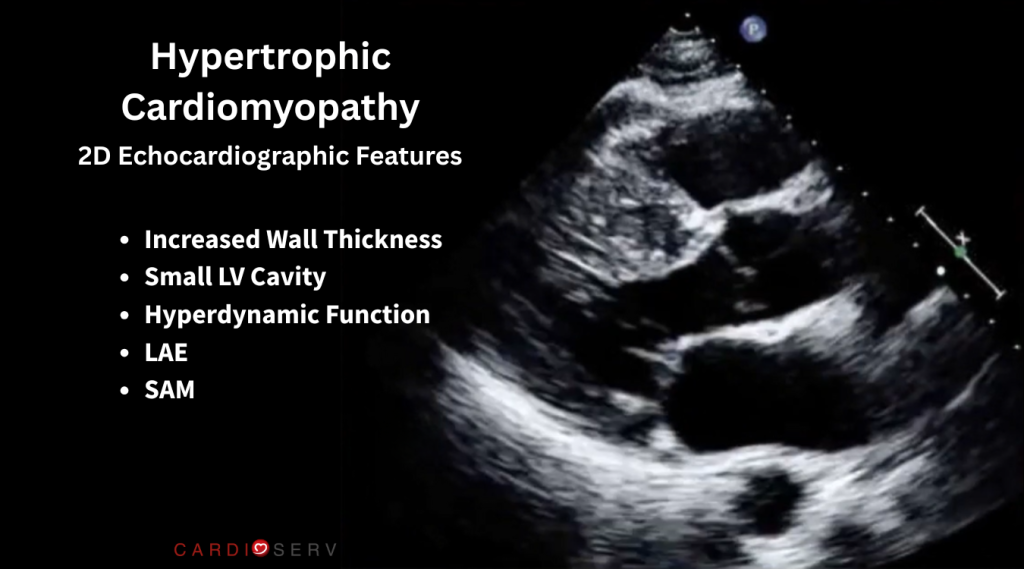
When scanning a patient, HCM should immediately come to mind when you see:
- Increased wall thickness (especially septal)
- Small LV cavity
- Hyperdynamic function (often EF 70%+)
- Enlarged left atrium
- SAM
- Possible turbulence in the LVOT
But recognition doesn’t stop there.
As a sonographer the moment you suspect HCM, your mindset should shift:
Ask yourself, “How can I tease this out so the physician clearly understands what I’m seeing?”
That single shift in mindset, from passive imaging to intentional investigation, is what separates a routine study from a diagnostic one, and can have a significant impact on patient outcomes.
Time to Pause
Imagine you’re scanning a patient with what appears to have LVH.
- The walls are thick and the basal septum appears even thicker.
- The LV cavity appears small
- LV function is hyperdynamic
- The left atrium is enlarged
- Turbulent flow in the LVOT
This is the moment to pause.
Because this is where HCM is either suspected and explored more in depth, or its missed.
The Importance of Accurate Measurements
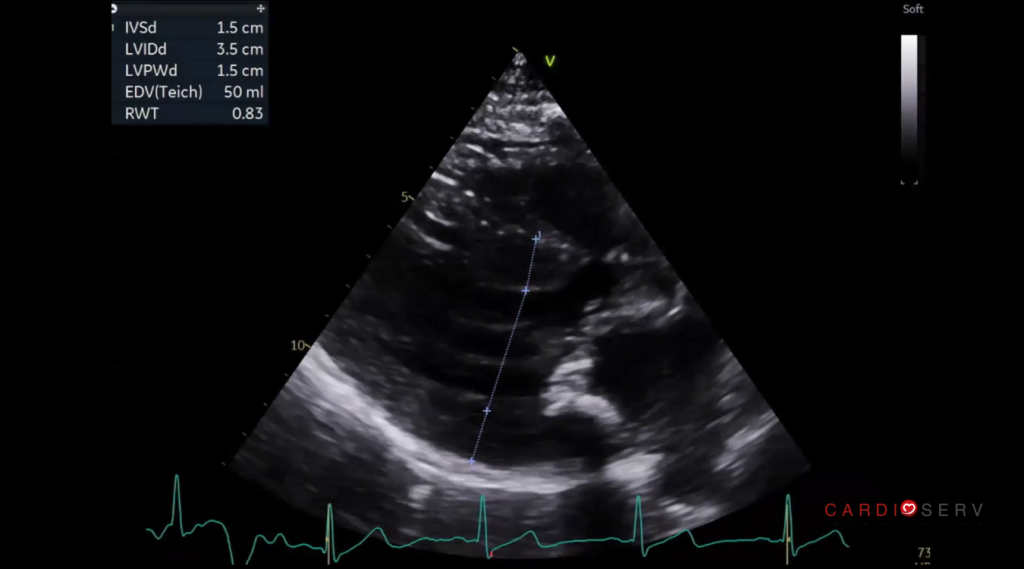
Once HCM is suspected, precision is critical. Even small errors, especially in wall thickness, can change how the study is interpreted.
Key considerations:
- Avoid including right ventricular structures in septal measurements
- Use multiple views (long and short axis)
- Identify the thickest segment, not just the basal septum
- Clearly document the pattern of hypertrophy
Probe position also matters. A slight shift in the intercostal space can improve alignment and help avoid RV inclusion.
SAM: A Critical Finding You Can’t Miss
Systolic anterior motion (SAM) of the mitral valve is a hallmark feature of obstructive hypertrophic cardiomyopathy and a key mechanism of left ventricular outflow tract obstruction. Failure to take the time to systematically assess for it may contribute to underrecognition of dynamic LVOT obstruction.
Instead of the mitral valve remaining in its expected position during systole, you’ll see:
- A distinct anterior motion toward the septum
- Sometimes even obvious septal contact
To consistently identify SAM utilize M-mode both on the mitral valve and on the aortic valve:
It’s true that many labs no longer rely heavily on M-mode, it can be especially helpful here.
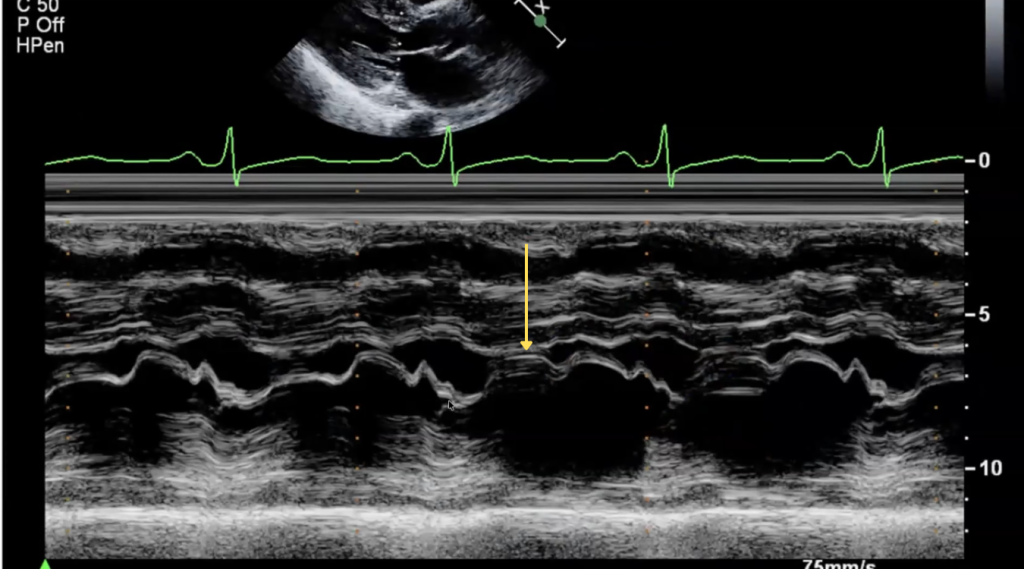
SAM on Mitral Valve M-Mode
- Place your M-mode cursor through the mitral valve tips in the parasternal long-axis (PLAX) view
- Focus specifically on systole and look for anterior motion of the mitral valve toward the septum
SAM on Aortic Valve M-mode
- Place your M-mode cursor through the aortic root and aortic valve leaflet tips in the parasternal long-axis (PLAX) view.
- On M-mode, the aortic valve opens normally, then partially closes halfway through systole before reopening, caused by a sudden decrease in flow due to obstruction.
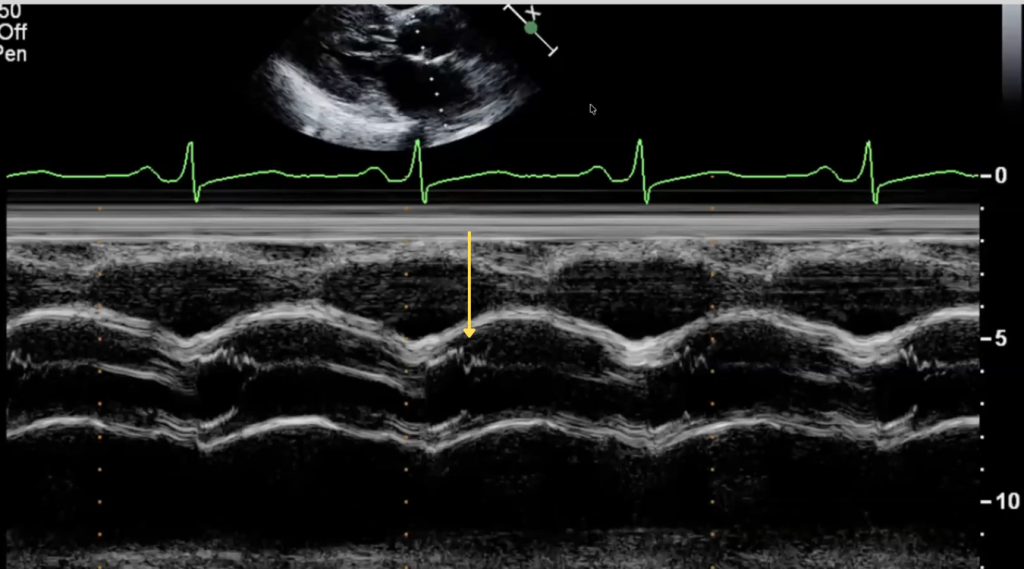
These simple steps can make a subtle finding much more obvious and reproducible.
So what actually causes this mitral leaflet motion that leads to obstruction?
What Causes LVOT Obstruction in HCM?
Like many things in echocardiography, understanding the “why” can help you recognize the “what.”
LVOT obstruction in HCM is often the result of a “perfect storm”:
- A thickened septum
- A hyperdynamic ventricle
- A mitral valve that is positioned or moves abnormally
These factors alter flow within the left ventricle, pushing the mitral valve into the LVOT and creating dynamic obstruction.
It’s important to note, however, that this is not a fixed obstruction.
HCM Is Dynamic. Not Static
One of the biggest mistakes in evaluating HCM is treating it like a fixed condition.
It’s not.
LVOT obstruction can change dramatically with:
- Preload
- Contractility
- Position
- Exercise
- Valsalva maneuver
A patient with a normal or mild gradient at rest can develop a severe gradient within seconds under the right conditions.
This is exactly why resting images alone are often not enough. As part of the complete assessment of HCM we must try and “unmask” any significant gradient. We can do this by asking the patient to perform valsalva.
In part 3 of the HCM series we’ll go into detail about how to use goal directed valsalva to easily evaluate the severity of the obstruction.
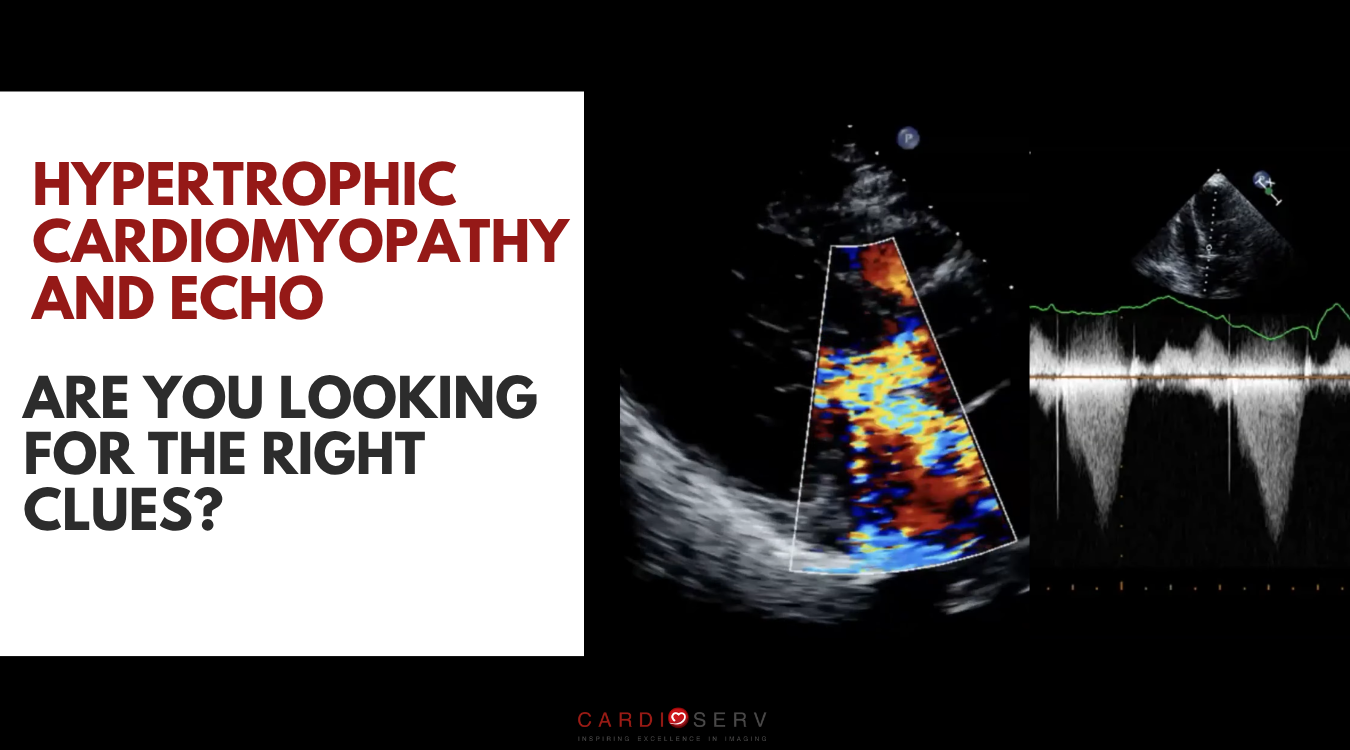
Clues You Should Not Ignore in Obstructive HCM
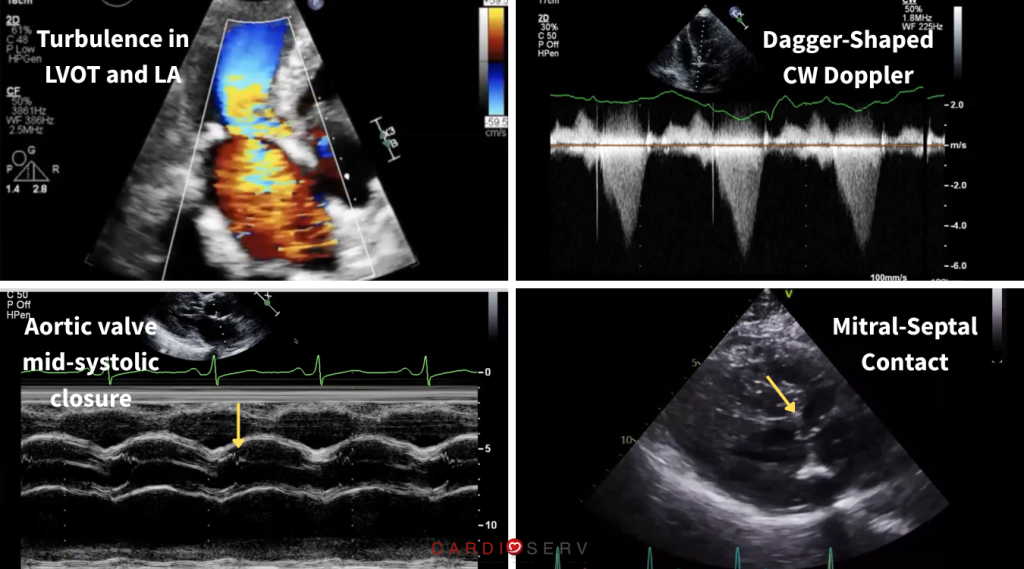
There are several subtle findings that can support the diagnosis of obstructive HCM:
- Turbulence in two directions (LVOT and left atrium)
- Late-peaking (dagger-shaped) CW Doppler waveform in the LVOT
- Mitral-septal contact
- Aortic valve mid-systolic closure (M-mode notch)
These are not isolated findings, they are pieces of a larger picture.
When they come together, they tell a much clearer story.
Think Before You Doppler
One of the most valuable practical things we can do at the patients bedside is to be intentional about Doppler placement.
Use your 2D and color Doppler first. Then decide where the Doppler cursor should go.
An important part of the HCM study is to differentiate true LVOT obstruction flow from mitral regurgitation. These two Doppler signals can look a lot alike, so it’s important to be intentional about cursor placement.
Too often, sonographers:
- Chase signals
- Sweep blindly with PW or CW Doppler
- Miss the true origin of flow
Instead:
- Identify turbulence with color
- Understand flow direction and differentiate the source
- Then position Doppler intentionally
- Label images so the reading physician can follow your thought process
These small adjustments can dramatically improve accuracy.
Why Standardization Matters
With increasing focus on HCM, consistency across labs is more important than ever.
HCM diagnosis should not depend on which sonographer performs the exam, which lab it’s done in, or which physician reads it.
This goes beyond individual skill.
It requires:
- Standardized protocols
- Consistent techniques
- Reproducible measurements
In larger organizations, this takes intentional effort.
What This Means for Your Next Study
When HCM is suspected, your approach should change.
In your next study:
- If you see asymmetric thickening → look for SAM
- If the cavity appears small and hyperdynamic → assess for LVOT turbulence
- If turbulence is present → confirm jet direction before placing Doppler
- If findings don’t fully align → pause and reassess before moving on
The goal isn’t to scan faster and get through the study, it’s to recognize when a routine study is no longer routine and adjust accordingly.
What Comes Next
Recognizing HCM is only the first step.
One of the most common and clinically important challenges is distinguishing LVOT obstruction from mitral regurgitation, especially when Doppler signals overlap.
In part 2 of this series, we’ll break this down step-by-step so you can confidently identify each pattern and avoid one of the most common sources of error in HCM echocardiography.
Ready to Go Even Deeper?
If you’ve ever looked back at a study and wondered if you missed HCM, or want to see exactly how these findings appear in real patients—this is where it comes together.
In this free webinar, you’ll see:
- Real case examples of HCM in the echo lab
- Clear demonstrations of SAM and LVOT obstruction
- Step-by-step breakdowns of how to scan and interpret these findings
- Differences between obstructive and non-obstructive HCM
- Access to Free downloadable resources for you and your lab
Final Thoughts
HCM isn’t rare, and it’s not hiding. It’s present in studies we’re already performing. It just isn’t always obvious.
With the right mindset and approach, it becomes much easier to recognize.
Because more often than not, it’s not that we’re not seeing it, we’re just not recognizing it for what it is.

Don Gerig, RDCS, Medical Education Manager
Connect on LinkedIn